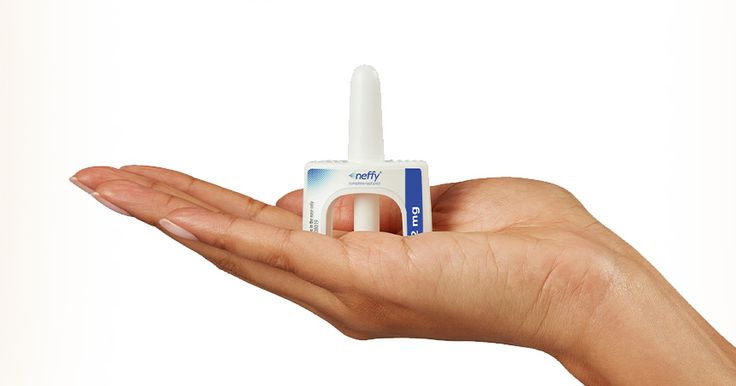
FDA Approves neffy (epinephrine nasal spray) Needle-Free Treatment for Type I Allergic Reactions, Including Anaphylaxis
SAN DIEGO, Aug. 09, 2024 (GLOBE NEWSWIRE) -- ARS Pharmaceuticals, Inc. (Nasdaq: SPRY), a biopharmaceutical company dedicated to empowering at-risk patients and caregivers to better protect patients from severe allergic reactions that could lead to anaphylaxis, today announced that the U.S. Food and Drug Administration (FDA) approved neffy® (epinephrine nasal spray) 2 mg for the treatment of Type I Allergic Reactions, including anaphylaxis, in adults and children who weigh ≥30 kg (66 lbs.). The approval represents the first significant innovation in the delivery of epinephrine in more than 35 years and is the first and only needle-free treatment option for patients and families living with severe allergic reactions.
“Until today, patients with severe allergic reactions, including anaphylaxis, only had one treatment option – an often painful and anxiety-inducing needle injection of epinephrine. In some cases, patients would delay or not administer the life-saving treatment at the onset of symptoms, increasing the risk for a severe reaction or negative outcomes requiring additional emergency medical treatment,” said Thomas B. Casale, M.D., Professor of Medicine and Pediatrics and Chief of Clinical and Translational Research in the USF Health Morsani College of Medicine's Division of Allergy and Immunology at the University of South Florida in Tampa, Florida. “FDA approval of neffy means that patients with severe allergies finally gain a long-awaited, needle-free, easy-to-carry epinephrine delivery method that has the potential to reduce time to administration, which can lead to better clinical outcomes and improvements in quality of life for patients and their caregivers.”
Type I allergic reactions, including those caused by food, medications, and insect bites, can lead to life-threatening anaphylaxis and result in an estimated 500,000 emergency room (ER) visits each year, with nearly 60% of such patients reported to not have received epinephrine prior to visiting the ER.
“This approval marks a watershed moment in addressing an unmet medical need for people with Type I allergies – a treatment alternative that avoids the need to inject epinephrine with a needle, which can be fraught with anxiety and fear for many,” said Richard Lowenthal, Co-Founder, President and Chief Executive Officer, ARS Pharmaceuticals. “Epinephrine treatment is only effective if available, readily usable, and administered appropriately. Our team has worked tirelessly to create an easy-to-carry, easy-to-use, needle-free device that offers peace of mind to patients and caregivers by enabling them to administer epinephrine quickly and confidently when needed. We thank the FDA staff for their partnership and support in the development of neffy, and are deeply grateful to the severe allergy community, including advocates, patients, parents, and healthcare professionals, for their contributions to the development of neffy as an important, life-saving treatment.”
The approval of neffy is based on data from five primary registration studies with a 2 mg intranasal dose of epinephrine. These primary clinical trials were supported by numerous pilot and supportive studies. neffy met all defined clinical endpoints and its pharmacokinetic (PK) and pharmacodynamic (PD) data were within the range of approved epinephrine injection products. These data included single- and twice-dosed studies in healthy adults, with self-administration and caregiver administration in Type I allergy patients, in pediatric patients ≥30 kg (66 lbs.) as well as in those with allergic rhinitis (congestion and runny nose). Adverse events in neffy clinical trials were generally mild in nature without any meaningful nasal irritation or pain, and no serious adverse events were reported in any clinical study.
“Anyone who has experienced or witnessed an anaphylaxis reaction knows it can be very stressful deciding when to inject epinephrine to themselves or a child and often delay,” said Dr. Jonathan Spergel, Chief of the Allergy Program at Children's Hospital of Philadelphia. “We know that earlier administration is better, and for many, the needle is a barrier that causes dangerous hesitation. That is why the field has long pursued an effective treatment approach that does not require an injection.”
Prioritizing Patient Access
As part of its commitment to access and affordability for all patients who need neffy, ARS Pharma plans to offer robust programs to support patient access. For people with severe allergic reactions who have commercial insurance that covers neffy, ARS Pharma is committed to limiting what most commercially insured patients pay to just $25 for each filled prescription of two single-use neffy devices via a co-pay savings program. For eligible patients without insurance coverage, situations in which a health plan does not yet cover neffy, or if a patient faces high out-of-pocket costs due to a high-deductible plan, ARS Pharma will offer a cash price of $199 for two doses of neffy via BlinkRx, a digital pharmacy service that is available to patients at no added cost. This cash price is also available for patients who use retail pharmacies via GoodRx, a free-to-use website and mobile app, or whose insurance plans explicitly carve out coverage of neffy. In addition, for eligible U.S. residents who are uninsured or underinsured, meet certain eligibility criteria, and have exhausted all other options, the ARS Pharma Patient Assistance Program (PAP) will provide neffy at no cost.
According to Dr. Lou Garrison, Professor Emeritus in The Comparative Health Outcomes, Policy, and Economics (CHOICE) Institute in the School of Pharmacy at the University of Washington, “neffy’s value to society should be measured by more than its benefit to the individual at the moment they use it to stop an anaphylactic reaction; its value is also in the reassurance it offers to everyone from just carrying it, knowing it can protect them in an emergency. Like a fire extinguisher in the home, neffy offers peace of mind to the person carrying it and also to their family and friends, who are reassured that the health of a loved one is better protected. But the peace of mind for potential patients and the similar spillover to their caregivers are values that are too often overlooked in health economics analyses. A product like neffy offers substantial societal value that should be embraced by insurance plans aligning care and coverage for patients affected by severe allergic reactions.”
“We join other patient organizations in our enthusiasm for the significant clinical and societal value of neffy, as well as ARS Pharma’s commitment to making it accessible for everyone no matter their insurance coverage,” said Peter Rubin, Executive Director of No Patient Left Behind, a non-profit dedicated to healthcare reforms that ensure patients can access the medicines of today and tomorrow. “Patients should not have to go without treatment due to out-of-pocket costs and ARS Pharma’s co-pay assistance program will help with that. This FDA approval is groundbreaking for patients living with severe allergies because neffy offers a clear benefit as an effective, convenient nasal spray. Ultimately, it is beneficial for all insurers and employer plan sponsors to cover neffy with affordable co-pays so that patients in need can access this needle-free epinephrine alternative when appropriately prescribed. Until then, we appreciate ARS Pharma for offering comprehensive programs to broaden patient access.”
Patient Support Available Through neffyconnect
To help ensure affordability and access, the neffyconnect program provides patients, caregivers, and healthcare professionals (HCPs) with information about patient support programs, such as medication fulfillment services and financial support to guide their treatment journey, including navigating insurance requirements and potential barriers to access. neffyconnect will also provide families with helpful educational resources and tools. ARS Pharma expects the majority of payors to formalize their coverage decision policies during the next six months; in the interim, neffyconnect can help HCPs and patients access financial support and medication fulfillment services. For more information, visit www.neffy.com or call 1-877-myneffy (1-877-696-3339).
“We commend the FDA's decision to approve ARS Pharma’s neffy, which offers the food allergy community a nasal-spray epinephrine delivery device,” said Sung Poblete, Ph.D., RN, CEO of FARE. “This needle-free breakthrough should make it easier for food allergy patients and those around them to administer epinephrine in the event of an anaphylactic reaction. Importantly, we believe that innovation of this nature should also make it easier to have epinephrine more broadly available for use in the event of an emergency—in police cruisers, at restaurants, in airplanes, at hotels, at amusement parks, and in other areas where accidental exposures are possible. This is a win for the food allergy community.”
“Studies show adults and teens with food allergies often don’t carry their auto-injectors. This new technology may overcome the inherent reluctance of people to carry bulkier epinephrine with them everywhere,” said Lianne Mandelbaum, an advocate for people with food allergies and Founder of the non-profit No Nut Traveler. “Currently the FAA only requires planes to include vials of epinephrine, which have to be administered with syringes. This can take time, and time is of the essence when treating an allergic reaction for the best possible outcome. This becomes critical in the air where backup medical care is not accessible. A small, convenient needle-free device may not only be vital to those who carry it but extend a safety net to fellow passengers who are not otherwise prepared for a mile-high allergic reaction.”
Product Availability
neffy is expected to be available in the United States within eight weeks of FDA approval for patients who weigh 30 kg or greater (66 lbs.). In the interim, patients can visit www.neffy.com to register for product updates, including when telemedicine services will be available. ARS Pharma plans to file a supplemental NDA application with the FDA for neffy (epinephrine nasal spray) for children who weigh 15 to <30 kg by the end of the third quarter of 2024. In the European Union (EU), neffy received a positive opinion and recommendation for approval from the Committee for Medicinal Products for Human Use on June 27, 2024, with commercial launch in the EU expected in Q4 2024 following the expected grant of market authorization by the European Commission (EC).
The shelf life of neffy is 30 months and allows for temperature exposure up to 122°F (50°C), making it a potentially effective treatment if left in a car or outside for a length of time. If accidentally frozen, neffy can be thawed and administered.
For more information, visit www.neffy.com.
About Type I Allergic Reactions, including Anaphylaxis
Type I severe allergic reactions are serious and potentially life-threatening events that can occur within minutes of exposure to an allergen and require immediate treatment with epinephrine, the only FDA-approved medication for these reactions. While epinephrine auto-injectors have been shown to be highly effective, there are well published limitations that result in many patients and caregivers delaying or not administering treatment in an emergency situation. These limitations include fear of the needle, lack of portability, needle-related safety concerns, lack of reliability, and complexity of the devices. There are approximately 40 million people in the United States who experience Type I severe allergic reactions due to food, venom or insect stings. Of those, only 3.3 million currently have an active epinephrine auto-injector prescription, and of those, only half consistently carry their prescribed auto-injector. Even if patients or caregivers carry an auto-injector, more than half either delay or do not administer the device when needed in an emergency.
Dr. Jonathan Spergel, Chief of the Allergy Program at Children's Hospital of Philadelphia, is a paid consultant and serves as a member of the ARS Pharma Scientific Advisory Board. He was not involved in the neffy clinical trials.
About neffy®
neffy® is an intranasal epinephrine product for patients with Type I allergic reactions including food, medications, and insect bites that could lead to life-threatening anaphylaxis.
INDICATION AND IMPORTANT SAFETY INFORMATION FOR NEFFY (epinephrine nasal spray)
INDICATION
neffy 2 mg is indicated for emergency treatment of type I allergic reactions, including anaphylaxis, in adult and pediatric patients who weigh 30 kg or greater.
IMPORTANT SAFETY INFORMATION
It is recommended that patients are prescribed and have immediate access to two neffy nasal sprays at all times. In the absence of clinical improvement or if symptoms worsen after initial treatment, administer a second dose of neffy in the same nostril with a new nasal spray starting 5 minutes after the first dose.
neffy is for use in the nose only.
Advise patients when to seek emergency medical assistance for close monitoring of the anaphylactic episode and in the event further treatment is required.
Absorption of neffy may be affected by underlying structural or anatomical nasal conditions.
Administer with caution to patients who have heart disease; epinephrine may aggravate angina pectoris or produce ventricular arrhythmias. Arrhythmias, including fatal ventricular fibrillation, have been reported, particularly in patients with underlying cardiac disease or taking cardiac glycosides, diuretics, or anti-arrhythmics.
The presence of a sulfite in neffy should not deter use.
neffy may alter nasal mucosa for up to 2 weeks after administration and increase systemic absorption of nasal products, including neffy.
Patients with certain medical conditions or who take certain medications for allergies, depression, thyroid disorders, diabetes, and hypertension, may be at greater risk for adverse reactions.
Epinephrine can temporarily exacerbate the underlying condition or increase symptoms in patients with the following: hyperthyroidism, Parkinson’s disease, diabetes, renal impairment. Epinephrine should be administered with caution in patients with these conditions, including elderly patients and pregnant women.
Adverse reactions to neffy may include throat irritation, intranasal paresthesia, headache, nasal discomfort, feeling jittery, paresthesia, fatigue, tremor, rhinorrhea, nasal pruritus, sneezing, abdominal pain, gingival pain, hypoesthesia oral, nasal congestion, dizziness, nausea, and vomiting.
These are not all of the possible side effects of neffy. To report suspected adverse reactions, contact ARS Pharmaceuticals Operations, Inc. at 1-877-MY-NEFFY (877-696-3339) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
For additional information on neffy, please see Full Prescribing Information at www.neffy.com.
About ARS Pharmaceuticals, Inc.
ARS Pharma is a biopharmaceutical company dedicated to empowering at-risk patients and caregivers to better protect patients from severe allergic reactions that could lead to anaphylaxis. The Company is commercializing neffy®, an epinephrine nasal spray product for patients with Type I allergic reactions, including food, medications, and insect bites that could lead to life-threatening anaphylaxis. For more information, visit www.ars-pharma.com.
Forward-Looking Statements
Statements in this press release that are not purely historical in nature are “forward-looking statements'' within the meaning of the Private Securities Litigation Reform Act of 1995. These statements include, but are not limited to, neffy’s potential benefits to patients and society; the timeline for neffy’s commercial availability; prices at which ARS Pharma plans to make neffy commercially available; ARS Pharma’s patient access prioritization for neffy; the platforms through which neffy will be accessible, including GoodRx and BlinkRx; the timeline by which payors will formalize their coverage decision policies; neffyconnect’s ability to help patients and HCPs access financial support and medication fulfillment services; ARS Pharma’s plan to file a supplemental regulatory application for a neffy 1 mg product for children 15 kg to <30 kg immediately after the approval of neffy 2 mg in the United States and/or Europe; the timeline for potential regulatory approval and commercialization of neffy in Europe; ARS Pharma’s expected cash runway; neffy’s shelf life and its effectiveness after being subject to extreme temperatures; and other statements that are not historical fact. Because such statements are subject to risks and uncertainties, actual results may differ materially from those expressed or implied by such forward-looking statements. Words such as “anticipate,” “plans,” “expects,” “will,” “potential” and similar expressions are intended to identify forward-looking statements. These forward-looking statements are based upon ARS Pharma’s current expectations and involve assumptions that may never materialize or may prove to be incorrect. Actual results and the timing of events could differ materially from those anticipated in such forward-looking statements as a result of various risks and uncertainties, which include, without limitation, the ability to maintain regulatory approval for neffy; results from clinical trials may not be indicative of results that may be observed in the future; potential safety and other complications from neffy; the labeling for neffy in any future indication or patient population; the scope, progress and expansion of developing and commercializing neffy; the potential for payors to delay, limit or deny coverage for neffy; the size and growth of the market therefore and the rate and degree of market acceptance thereof vis-à-vis intramuscular injectable products; ARS Pharma’s ability to protect its intellectual property position; and the impact of government laws and regulations. Additional risks and uncertainties that could cause actual outcomes and results to differ materially from those contemplated by the forward-looking statements are included under the caption “Risk Factors” in ARS Pharma’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2024, filed with the SEC on August 6, 2024. This document can also be accessed on ARS Pharma’s web page at ir.ars-pharma.com by clicking on the link “Financials & Filings.”
The forward-looking statements included in this press release are made only as of the date hereof. ARS Pharma assumes no obligation and does not intend to update these forward-looking statements, except as required by law.
Source: ARS Pharmaceuticals, Inc.
Posted: August 2024